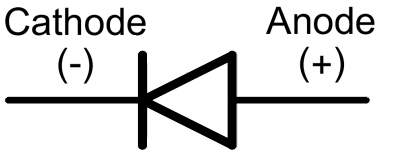 
He fundamentally defined his arbitrary orientation for the cell as being that in which the internal current would run parallel to and in the same direction as a hypothetical magnetizing current loop around the local line of latitude which would induce a magnetic dipole field oriented like the Earth's. The reference he used to this effect was the Earth's magnetic field direction, which at that time was believed to be invariant. His motivation for changing it to something meaning 'the East electrode' (other candidates had been "eastode", "oriode" and "anatolode") was to make it immune to a possible later change in the direction convention for current, whose exact nature was not known at the time. Previously, as related in the first reference cited above, Faraday had used the more straightforward term "eisode" (the doorway where the current enters). The use of 'East' to mean the 'in' direction (actually 'in' → 'East' → 'sunrise' → 'up') may appear unnecessarily contrived. In that paper Faraday explained that when an electrolytic cell is oriented so that electric current traverses the "decomposing body" (electrolyte) in a direction "from East to West, or, which will strengthen this help to the memory, that in which the sun appears to move", the anode is where the current enters the electrolyte, on the East side: " ano upwards, odos a way the way which the sun rises" (, reprinted in ). The word was coined in 1834 from the Greek ἄνοδος ( anodos), 'way up', by William Whewell, who had been consulted by Michael Faraday over some new names needed to complete a paper on the recently discovered process of electrolysis. In a cathode ray tube, it is the positive terminal where electrons flow out, i.e., where current flows in.Īn electrode through which current flows the other way (out) is a cathode.Note electrode naming for diodes is always based on the direction of the forward current (that of the arrow, in which the current flows "most easily"), even for types such as zener diodes or solar cells where the current of interest is the reverse current. In a diode, it is the positive terminal at the tail of the arrow symbol, where current flows into the device.In a recharging battery, or an electrolytic cell, the anode is the positive terminal, which receives current from an external generator.If the anode is composed of a metal, electrons which it gives up to the external circuit must be accompanied by metal atoms missing those electrions (cations) moving away from the electrode and into the electrolyte. At the anode, the current is continued internally by positive ions ( cations) moving into the electrolyte from the anode, i.e., away (surprisingly) from the more negative electrode and towards the more positive one (chemical energy is responsible for this "uphill" motion). Since this inwards current is carried externally by electrons moving outwards, the negative charge moving one way amounts to positive current flowing the other way. 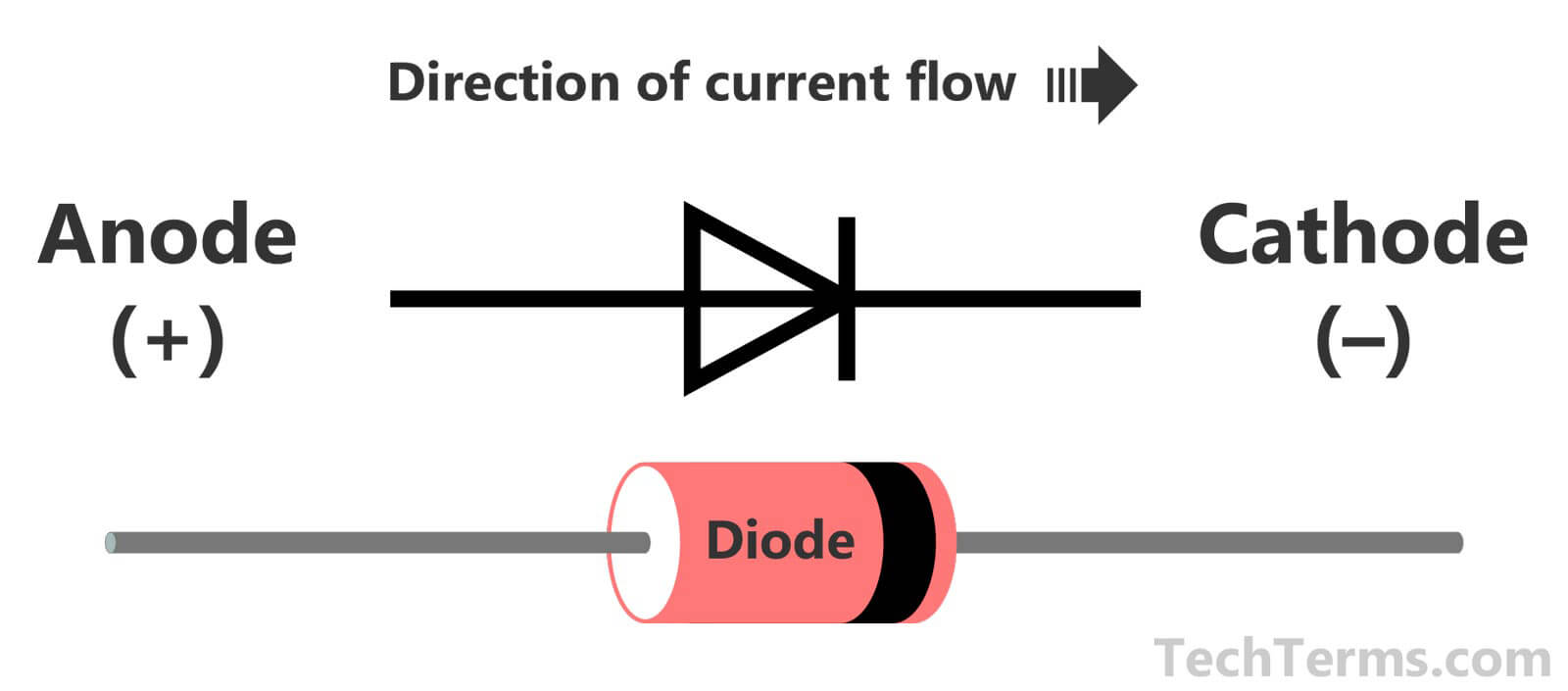 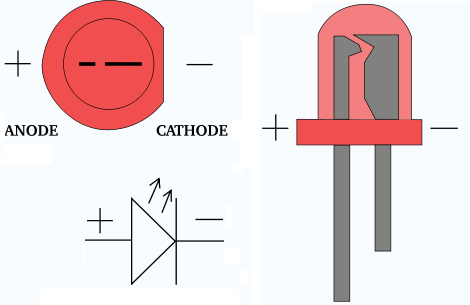
In a discharging battery or galvanic cell (drawing) the anode is the negative terminal, where conventional current flows in, and electrons out.To dispel a common misconception, often incorrectly inferred from the correct fact that in all electrochemical devices negatively charged anions move towards the anode and/or positively charged cations move away from it, anode polarity is not always positive but depends on the device type, and sometimes even in which mode it operates, as determined by the above current direction-based universal definition. Electrons flow in the opposite direction to the positive electric current. Mnemonic: ACID (Anode Current Into Device). Diagram of a zinc anode in a galvanic cell.Īn anode is an electrode through which (positive) electric current flows into a polarized electrical device.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
